@sean.a.hyde
I'm not really going to address or comment on the whole debate here regarding the use of technology versus titration methods for pool water testing. I understand both sides of the argument and I only know what is working well for me presently. I use both. Technology for day-to-day monitoring and titration as a backup occasionally to insure that the technology has not led me astray as evidenced by my pool math logs.
I would like to address your comments regarding the Atlas Scientific probes/sensors however and see if I can offer up some points for consideration.
1. As
@Katodude pointed out earlier, there are a handful of us here that are using the Atlas Scientific probes/sensors. As far as I'm aware, all with good success. That is not to say that there have not been "speed bumps" along the way, because in my case there were. But after negotiating some pitfalls that were due to my own erroneous assumptions and getting some very helpful assistance from other TFP members, I can honestly say that I am very pleased with the results now.
2. Personally, I am currently using six Atlas Scientific sensor/probes.
ORP
pH/Water Temp
EC
Air Temperature
Filter Pressure Transducer
Humidity/Dewpoint
Do I need all of these for pool chemical management? Heck no. I just like to play with the technology and see what I can make work for me (or not lol).
My ORP readings are merely an indicator for me because of my higher CYA levels (80ppm). But when I look at either my chemical management dashpanel or my Grafana dashboard and if I were to see an unusually low ORP value, it just tells me that I might want to double check my FC level (yes, using the Taylor) and to check my IntelliChlor output level. That is all I use it for really. That said, I agree with
@Katodude , that at some point those reliable amperometric free chlorine sensors are going to get better and cheaper and then I'll add one to my sensor manifold. If and when that occurs, maybe it might be possible through chemical management software to tie SWCG output to the FC sensor readings.
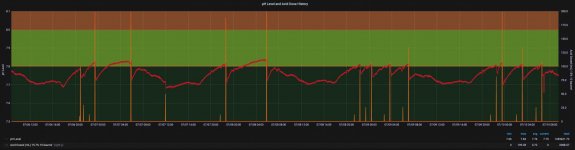
3. pH - As far as your statement regarding "periodic drift during the day/night". Have you considered that may only be due to the differences in pool water temperature between day and night? I have a play/lap pool (only 5.5' in the middle), so this time of year, my pool water temperature fluctuates an average of of 10-15 degrees F between night and day.
Since water temperature has a direct impact on pH readings, my pH can fluctuate an entire .1 between night and day. At this time of the year, typically this will cause my pH to drop to an average of 7.7 during the hottest part of the day when my pool water temp is nearly 90-93 degrees and rise to above 7.8 when the pool water temperature averages around 80 degrees in the early morning hours. The pH would rise even further at night but when it reaches 7.8 (my MA dosing setpoint) and is trending up, then the chemical management software that I am fortunate enough to use, slaps it back down (doses muriatic acid), as evidenced by the following graph.
View attachment 353681
As you can see, my pH decreases as the water temp increases and vice versa. So it may be entirely possible that those periodic drifts between night and day that you speak of, are merely due to water temperature fluctuations and not a problem with your Atlas probe.
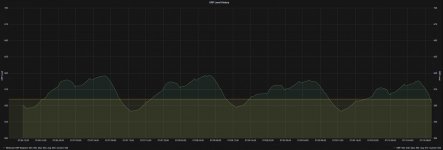
Although for a different reason of course, my ORP also fluctuates between night and day as well, as you can see from the graph below. That is also normal..
View attachment 353682
Which brings me to my last point. I'm not really sure what type of software that you are using that interprets your Atlas Scientific probes/sensors, but my own opinion is that it can play a major role in how well your data is presented etc. Just a thought.
I merely wanted to offer up a different perspective that might show that using the Atlas Scientific sensors and probes provides much more than just a "clock" to some of us. I hope you have better luck with your Atlas Scientific sensors and like I said, there are a handful of us here that are having success with them and I'm pretty sure that any one of us would be happy to assist you to get to the point where you might want to be with them.

Thanks,
r.