I am curious to know what the minimum threshold is so I can determine the correct buffers for the situations specific to my pool.
If the CDC requires 1ppm they are leaving room for some contingency.
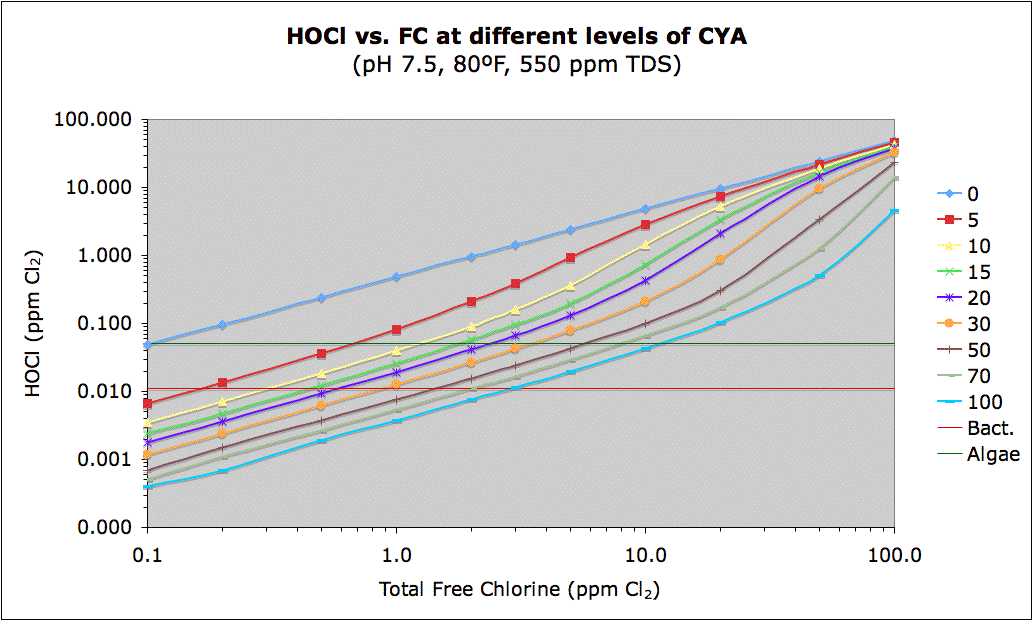
The above chart confirms my belief that CYA should only be used in situation where free chlorine can't be maintained.
I have no problem holding chlorine, I have a screened in lanai that blocks 30% to 50% of UV rays and my pool is heavily shaded by my home and surrounding trees. It looks like I can maintain free chorine at 0.5 ppm and have plenty of buffer if the pool has no load. My automation system is very precise and highly responsive so holding 0.5 ppm is achievable. I see no reason to work keep a sanitized and balanced pool at 3ppm if the sun is just going to burn off the chlorine. If I start to see algae I can just SLAM and increase my set point.
To me one of the most interesting things about Chem Geek's post is:
"Note that the red in the linked chart above represents a cutoff of 0.011 ppm HOCl which roughly corresponds to the 650 mV ORP level that the U.S. and WHO set as the minimum required for disinfection."
With my automated acid feeder I can keep my PH between 7.25 and 7.35 where the ratio of HOCl is higher. So with my liquid chlorine is added it is highly effective without any CYA. At 0.5 ppm my HOCl is 29x the WHO requirement. I could probably get away with keeping free chlorine between 0.2 and 0.4 and still have plenty of buffer. I have my Becsys set to feed chorine based on ORP so that metric will provide additional buffering.
Richard also states: "perhaps hypochlorous acid (HOCl) is less susceptible to breakdown from sunlight than hypochlorite ion (OCl-). This implies that having a pool at lower pH results not only in more disinfecting chlorine, but has the chlorine last longer (though the effect may not be very strong from, say, 7.8 to 7.2)". I noticed early on that my pool looked much better when the PH was closer to 7.0... This is an theory I plan to test on my own pool this summer.