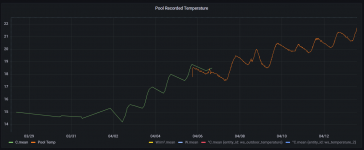
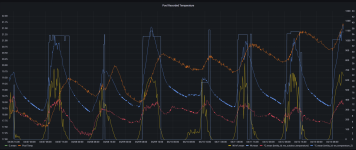
At this stage should I continue to try to lower the total alkalinity using muriatic/hydrochloric acid additions, then aeration to bring the ph back up? I don't fully understand if this process will work when it appears the water hardness appears to be the only contributor to the high TA?
Even though Hardness (CH) and TA are both measured in the unit ppm CaCO3, they are still different things.
Having a CH of 100ppm means you have the same amount of CA++ ions in the water as you would get from adding 100mg of CaCO3 to 1l of water and dissolving into the water as Ca++ and CO3-- (Carbonate) ions. That doesn't necessarily mean that the Ca-- ions got into your water by adding calcium carbonate, it's just an equivalent.
In the same way a TA of 100ppm means that you have the same TA in the water as you would get from adding 100mg of CaCO3 to 1l of water and dissolving into the water as Ca++ and CO3--. Here it's basically giving the equivalent amount of Carbonate ions (CO3--) creating the same TA as you have in the water.
Depending on the pH, some of the CO3-- (which has a double contribution to TA because of the two charges) will change into HCO3- (which has only a single contribution to TA, but because in the process one H+ got consumed, which has a negative contribution to TA, the net TA is unaffected) and into H2CO3 (which has no contribution to TA, but the process removed another H+). The H2CO3 actually splits mostly up into CO2 (which drives CO2 outgassing) and water (H2O).
Then, in a pool, there is also a contribution of CYA to TA. But all of these TA contributions are basically measured as an equivalent amount of added CaCO3 that would yield the same TA in the form of CO3--.
To calculate the Calcite Saturation Index (CSI), which tells us if water is scaling (CSI>0) or aggressive to plaster surfaces (CSI<0), we are interested in the amount of CO3-- in the water, which can be calculated from TA considering pH and CYA.
When both, the amount of CO3-- and Ca++, is too high (technically it's the product of the two), then the water is "oversaturated", and some Ca++ and CO3-- will form CaCO3 and fall out of solution as scale. If the product of the two is too small, then the water is undersaturated with CaCO3 and can draw some Calcium out of plaster surfaces to reach saturation.
Therefore, you don't want both CH and TA high. Someone with low CH can afford higher TA in terms of CSI.
But independent from CSI, high TA always drives pH drifting upwards by CO2 outgassing. That's why we recommend to maintain lower TA to slow down pH drifting up.
That was a lot of stuff, hope I didn't overwhelm you with that...