Maybe one last point on chemistry. It takes usually some time to get your head around the higher FC levels we recommend here, so a little background on that.
When adding chlorine to water you end up with hypochlorous acid and hypochlorite ion. Without any CYA in the water, this is what shows up as FC in the chlorine test. It is the hypochlorous acid part that we are interested in, this is what kills bacteria and algae, and also what makes chlorinated water feel "harsh" and wears out swim suits over time. We also like to call this "active" chlorine.
When you add CYA, things change a bit. About 95% of the chlorine is now attached to CYA. The good thing about that is that it is protected from UV light there, which is why we use it in the first place. The downside is that chlorine that's attached to CYA has no more algae/bacteria killing powers. Unfortunately, it still shows up as FC in the test. What needs to be done is to increase FC to make sure that there is enough active chlorine. Turns out that same ratios of FC/CYA contain same amounts of active chlorine. That means when doubling CYA, you also need to double FC.
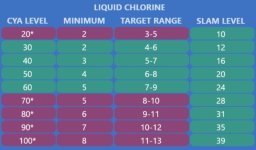
To give you some numbers:
SLAM FC (for example FC 12 for CYA 30, or FC 24 for CYA 60) contain the same amount of active chlorine than FC 0.64 without any CYA in the water.
That's right, FC 0.64 is enough to SLAM a pool without CYA. Problem with that is that you'd lose most of that FC to UV in no-time, rather than getting it to work on algae.
The target FC ranges in our recommended
FC/CYA Levels, are equivalent to something like FC 0.1 to 0.15 without CYA.
This is for example reflected in codes for public pools in Europe, where FC (without CYA) is limited to 0.6. This gives a great swimming experience, but you need professional equipment to maintain these levels, that would not work in a residential backyard pool.
If used properly, CYA does the magic in our backyard pools. It protects chlorine from FC and provides a reservoir of chlorine without the water being aggressive. As your active chlorine goes down from UV, killing bacteria and algae, or doing its job on the stuff that swimmers leave behind, more active chlorine gets released from CYA.
Chlorine is not locked into the CYA, that's not how chemistry works (so delete terms like "chlorine lock" that you may have heard from pool stores from your vocabulary). There is always an equilibrium, at given levels of FC, CYA and pH you have well defined levels of active chlorine and chlorine attached to CYA. If you suddenly remove all of the active chlorine (let's say due to kids doing what kids do in water), then more active chlorine gets released from the "reservoir" of chlorinated CYA until everything is back in equilibrium - this happens to our "eyes" pretty much instantaneously.
Also turns out that this magic includes that with CYA the active chlorine levels are not very dependant on pH (without CYA they are, most of what shows up as FC would be hypochlorite ion at high pH which is far less powerfull than hypochlorous acid - that is where the urge from pool stores to always bring pH down to 7.2 comes from). So, with CYA in the water it is absolutely fine to maintain pH more around 7.8 to 8.0, which will (in combination with TA around 70ish) lead to a much more stable pH without having to add acid so often.
All of these chlorine / CYA equilibrium thingies have been first published in science journals in the late 1960ies. Unfortunately, the pool industry has decided to widely ignore that for decades.
I hope I didn't dive too deep here, but I wanted to give you some reassurance that we are not just making these things up. We are just applying scientific learnings to pool care.